Return to Appendices
APPENDIX I -- THE COMPOSTING HANDBOOK
ODORANTS -- CHEMICAL COMPOUNDS THAT CAN CONTRIBUTE TO COMPOSTING ODORS
Robert Rynk
Latest revision: December 10, 2021
At its most basic, composting is the decomposition of complex organic substances. As these complex substances decompose, they produce secondary organic compounds, at least temporarily. Some of these secondary compounds are volatile; that is, they can evaporate into the surrounding air before they further decompose. A number of these volatile secondary compounds are also odorants. Odorants are chemical compounds that have a recognizable odor. The odors associated with some odorants are offensive, at least to some people.
The odors that occasionally plague composting are rarely the result of the overabundance of a single odorant. Rather, odorants usually coexist and interact with one another to produce diverse aromas. Nevertheless, a particular odorant, or class of odorants, may dominate the overall odor. At the very least, a particular odorant may lend its character to a more general odor. Therefore, it can be helpful to appreciate the compounds that can contribute to the odor of composting, and their characteristics. An odor wheel is one tool that can help zero-in on the odor character that individual odorants might be contributing to an overall odor. Figure 1, reprinted from BioCycle magazine, is an example (Mendrey 2014; https://www.biocycle.net/the-compost-odor-wheel/).
Composting produces numerous volatile compounds that are considered odorants. They are commonly identified by categories of similar chemicals. The primary categories identified with organic materials are ammonia, amines, indoles, mercaptans, organic sulfides, hydrogen sulfide, volatile fatty acids (VFAs), terpenes, alcohols, ketones and aldehydes (Goldstein, 2002). The first three in this list are compounds that contain nitrogen (N). Mercaptans, organic sulfides, and hydrogen sulfide, are sulfur-based compounds. Volatile fatty acids (VFAs) are most closely associated with carbohydrates and lipids (i.e., fats and oils). The remaining groups are natural organic compounds with volatile members.
Table 1, which is reproduced here from Chapter 12, summarize the chemical compounds that are most often responsible for odors at composting facilities. The general categories are described below (see CalRecycle 2007 for more detail; https://www2.calrecycle.ca.gov/Publications/Details/1241.).
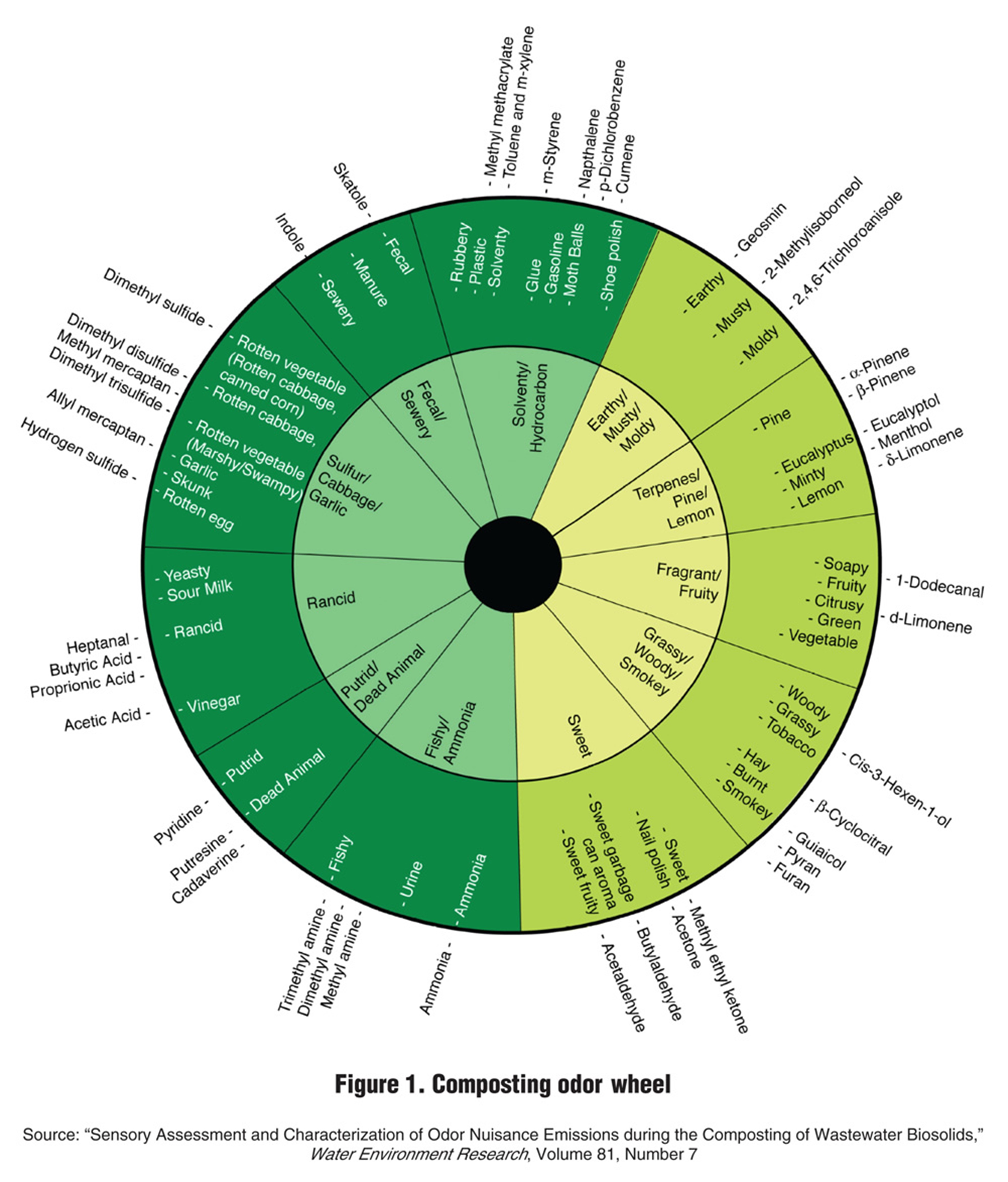
Reprinted with permission of BioCycle Connect.
Table 12.1. Common odorants released during handling and decomposition of organic materials. Adapted from CalRecyle (2007) and compiled from various sources including Brant and Elliott (2004), Chiumenti et al., (2005); Williams and Miller (1993); Wilmink and Diener (2001); and Epstein (1997) and Engineering Toolbox, (2006).
|
Compound Name
|
Odor Descriptors
|
Chemical Formula
|
Vapor Pressure at 20 or 25°C (mmHg)
|
|
|
Volatile Fatty Acids (VFAs)
|
|
Formic acid
|
Biting, pungent
|
HCOOH
|
42.8
|
|
|
Acetic acid
|
Vinegar-like, pungent
|
CH3COOH
|
15.7
|
|
|
Propionic acid
|
Rancid, pungent
|
C2H5COOH
|
3.53
|
|
|
Butyric acid
|
Rancid butter, body odor
|
C3H7COOH
|
1.65
|
|
|
Valeric acid
|
Unpleasant, sweat
|
C4H9COOH
|
0.1
|
|
|
Isovaleric acid
|
Rancid cheeses
|
(CH3)2C2H3COOH
|
0.554
|
|
|
Caproic acid (hexanoic acid)
|
Pungent
|
C5H11COOH
|
0.2
|
|
|
Volatile Nitrogen Compounds: ammonia, amines, indoles
|
|
Ammonia
|
Pungent, sharp, irritating
|
NH3
|
7510
|
|
|
Putrescine
|
Putrid, nauseating
|
NH2(CH2)4NH2
|
4.12
|
|
|
Cadaverine
|
Putrid, decaying flesh
|
NH2(CH2)5NH2
|
1.0
|
|
|
Methylamine
|
Putrid, fishy, rotten fish
|
CH3NH2
|
1810
|
|
|
Dimethylamine
|
Fishy, rotten fish
|
(CH3) 2NH
|
1520
|
|
|
Trimethylamine
|
Fishy, pungent
|
(CH3)3N
|
2650
|
|
|
Ethylamine
|
Ammonia-like, irritating
|
C2H5NH2
|
|
|
|
Indole
|
Fecal, nauseating
|
C6H4(CH2)3NH
|
0.012
|
|
|
Skatole
|
Fecal, nauseating
|
C9H9N
|
0.015
|
|
|
Volatile Sulfur Compounds: hydrogen sulfide, mercaptans, organic sulfides
|
|
Hydrogen sulfide
|
Rotten egg
|
H2S
|
15600
|
|
|
Methyl-mercaptan
|
Pungent, rotten cabbage, skunk, garlic
|
CH3SH
|
1510
|
|
|
Ethyl-mercaptan
|
Rotten cabbage, leek-like
|
C2H5SH
|
529
|
|
|
Carbon disulfide
|
Disagreeably sweet, rotten pumpkin
|
CS2
|
360
|
|
|
Dimethyl sulfide
|
Sulfurous, rotten cabbage,
|
(CH3)2S
|
502
|
|
|
Dimethyl disulfide
|
Putrid, sulfurous
|
(CH3)2S2
|
28.7
|
|
|
Terpenes
|
|
α-Pinene
|
Sharp, turpentine
|
C10H16
|
4.75
|
|
|
Limonene
|
Sharp, lemony
|
C10H16
|
1.98
|
|
|
Ketones and Aldehydes
|
|
Phenol
|
Medicinal
|
C6H5OH
|
0.38
|
|
|
Acetone
|
Pungent, solvent
|
CH3 COCH3
|
225
|
|
|
Methyl ethyl ketone (Butanone)
|
Sweet, solvent
|
CH3 COCH2 CH3
|
72
|
|
|
Formaldehyde
|
Acrid, medicinal
|
H2CO
|
3883
|
|
|
Acetaldehyde
|
Green, sweet, fruity
|
CH3CHO
|
900
|
|
1Nitrogen Compounds
The decomposition of proteins and other organic substances generates a number of nitrogen-based compounds that are volatile and odorous. Ammonia is by the far the most prominent and important or these in terms of nitrogen (N) loss. Because of their offensive odors, two groups of organic nitrogen compounds are also notable -- amines and indoles. Whenever N is abundant, and especially where C:N ratios are low (< 25:1), odorous nitrogen compounds are likely to be emitted. These N compounds develop inherently as proteins decompose. Under aerobic conditions they continue to decompose and disappear relatively quickly, with the exception of ammonia.
To reduce the formation of nitrogen-based odorants (e.g. ammonia, amines) and also VFAs, the C:N ratio of the feedstock mix should be high, certainly above 25:1, and preferably well above 30:1. In addition, much of the C that accounts for the C:N ratio should be available C. If the C is largely from wood-based feedstocks, the ratio of total C:N should be even higher. Available C helps microorganisms assimilate the N compounds that arise during decomposition, rather than volatilizing as ammonia or amines. A high C:N ratio also slows down the microbial activity, reducing the oxygen demand and deterring anaerobic conditions.
1.1 Ammonia
Ammonia and its pungent odor are present at composting facilities that handle feedstock mixtures with a high nitrogen content (e.g., with healthy proportions of manure, biosolids, fish, grass, etc.). Ammonia is emitted wherever high-nitrogen wastes are produced, stored or handled and the pH and/or temperature are elevated. It readily forms under both aerobic and anaerobic conditions. Ammonia is a distinct problem for composters handling very high nitrogen feedstocks such as poultry manure and fish waste. Otherwise, ammonia is primarily an odor concern at composting facilities. It is seldom associated with odor complaints that occur beyond the facility. Like H2S, ammonia tends to dissipate rapidly after it is emitted (Haug, 1993). Also, many people are familiar with the smell of ammonia and do not find it suspicious. Nevertheless, because of its ubiquitous nature, ammonia is a prominent odorant and the primary reason for N loss during composting.
1.2 Amines
Amines are foul smelling volatile organic nitrogen compounds, that result from the decomposition of proteins and amino acids. In chemical structure, some amines are similar to organic sulfur compounds. Some other amines that may contribute to odors include methylamine, ethylamine and trimethylamine. These amines tend to impart an odor that is often described as fishy and/or ammonia-like. One would intuitively expect them to be present and to affect the odor quality at facilities that handle highly degradable nitrogen-rich feedstocks like fish waste, animal mortalities, poultry manure, grass and biosolids.
1.3 Indoles
Two compounds in this group, indole and skatole, have a feces-like odor. Although these indoles have not been identified as major odorants at composting facilities, they may contribute a negative character to the general odor where manure and biosolids are handled., Bacteria in animal intestines produce indole and skatole. However, plants also produce indoles. Ironically, at low concentrations some indoles are partially responsible for the pleasant scent in some flowers and perfumes.
2 Sulfur Compounds
Sulfur (S) is a common to several of the more objectionable and recognizable odorants. Sulfur is relatively abundant in many composting feedstocks including some food residuals (e.g. onions), paper, manure, biosolids and gypsum (Miller,1993). Sulfurous compounds principally identified as odorous include hydrogen sulfide, mercaptans and organic sulfides (e.g. dimethyl disulfide).
Sulfur-base odorants are more likely to be immobilized, and less likely to volatilize, if a sufficient pool of C is available. Stable organic matter has a C:S ratio of approximately 100:1 suggesting that materials with lower C:S ratios harbor excess sulfur that could potentially volatilize. Therefore, one possible way to minimize emission of sulfur-based odorants is to establish a mix where C:S ratios are above 100:1. While there is little composting research supporting this practice, the principle is sound, and similar to the logic applied to C:N ratios (Miller 1993).
2.1 Hydrogen sulfide
Hydrogen sulfide (H2S) is a reduced and inorganic form of sulfur that produces a familiar rotten egg smell. While it can form at low oxygen concentrations (anoxic), the presence of H2S indicates anaerobic decomposition is taking place. Although H2S produces an offensive odor, and it can be detected at low concentrations, it does not appear to be a major source of odors at composting facilities. One possible reason is that H2S tends to dissipate and/or oxidize quickly in the environment (Walker and Gossett, 1999). Also, H2S is not volatile at pH levels above 8 (Das, 2000). Hydrogen sulfide is heavier than air and can accumulate in confined environments, raising the possibility of asphyxiation.
2.2 Mercaptans
Mercaptans, also termed thiols by chemists, are characterized by a strong odor and detectable at low concentrations (Haug, 1993). Naturally occurring mercaptans are responsible for the odors that skunks emit and the aromas of garlic and onions., Methyl mercaptan is used in low concentrations to impart an odor to natural gas. In decomposing organic matter, mercaptans result from the early steps of protein degradation (Miller, 1993). They can form aerobically but occur more abundantly under anaerobic conditions. Mercaptans have been identified as contributors to composting odors from several feedstocks, including food, paper processing residuals, biosolids and swine manure.
2.3 Organic Sulfides
Volatile organic sulfur compounds associated with odors include dimethyl disulfide (DMDS), dimethyl sulfide (DMS) and carbon disulfide (CS). These compounds have a strong offensive odor and are detectable at low concentrations. They are often cited as the dominant sources of odors from manure and biosolids. These sulfur compounds can form during aerobic or anaerobic decomposition persist under anaerobic conditions. Of these organic sulfides, DMDS appears to be the major odor source at biosolids composting facilities (Goldstein, 2002, Epstein, 1997).
3 Volatile Fatty Acids (VFA)
Volatile fatty acids (VFAs) are also referred to by the more general term volatile organic acids (VOAs). VFAs are “sour-smelling” compounds that develop as carbohydrates, fats and oils decompose. The most familiar VFA is acetic acid or vinegar. Other examples include formic, propionic, and valeric acids, which have odors descriptors like rancid and putrid. Butyric acid is sometimes associated with the characteristic smell of “garbage” (and also butter). Numerous VFAs can develop during decomposition, each with a characteristic odor. As VFAs form, the pH drops within the composting materials. If oxygen is abundant, VFAs readily decompose and the pH recovers. However, if oxygen is scarce, the pH remains low and VFAs accumulate and volatilize.
VFAs particular afflict composting facilities that handle food wastes because food is rich in organic compounds that quickly decompose. Yard trimmings composting facilities also can be troubled by VFAs, in part because of the relative scarcity of nitrogen- and sulfur-based odorants (except if grass is prevalent). However, VFAs are possible with almost any organic material, especially when anoxic conditions prevail.
4 Terpenes
Terpenes are common odorants that are naturally-produced by various plants, including lemons, geraniums, rose, mint, pine and eucalyptus (Haug, 1993). These aromatic compounds are liberated from the plant cells as the leaves, stems, bark and branches are shredded and turned during composted. Terpenes are prominent where woody materials are composted, such as brush and tree branches at green waste facilities, sawdust bedding in manure, and wood chip bulking agents used in composting biosolids (Epstein, 1997)
At low to moderate concentrations, the smell of terpenes is tolerable, even pleasant, to most people. However, at high concentrations, terpenes can present an bothersome odor. Also, when mixed with the soup of other odors of decomposing materials, they may add to the intensity of an overall disagreeable smell. At composting facilities, terpenes appear to be prominent where woody materials are composted, such as brush and tree branches at green waste facilities, sawdust bedding in manure, and wood chip bulking agents used in composting biosolids (Epstein, 1997)
5 Other Odorants
Odors can emanate from several other categories of common volatile organic compounds including ketones, aldehydes and alcohols, among others. Aldehydes and ketones are produced from the oxidation of different types of alcohols and further oxidize to form organic acids. Alcohols result from the oxidation of hydrocarbons, including fermentation. These types of compounds occur naturally as various organic substances degrade. In aerobic environments they tend to be short lived. Whether or not the odors from these compounds are deemed unpleasant depends on their concentrations, intensity, human sensitivity to these compounds and the presence of other odorous substances. In general, they are rarely a primary component of malodors at a composting site but can contribute negatively to overall odor character. In certain situations, or with specific materials, an alcohol, ketone or aldehyde may produce a detectable and dominant odor.
5.1 Alcohols
The formation and fate of alcohols during composting are similar to that of VFAs. When carbohydrates are abundant, their subsequent decomposition leads a decrease in pH and an accumulation of alcohols and organic acids. The low pH can retard further decomposition of both. With time (and oxygen), the pH increases and alcohols eventually decompose but, in the meantime, the material can emit the sour smell of alcohols, often commingles with VFAs. This situation is a typical of feedstocks that are rich in easily degradable carbohydrates (e.g., potato culls, some food wastes).
5.2 Ketones
Ketones such as acetone and methyl ethyl ketone (butanone) are quite volatile and have a “sweet” solvent-like odor. However, human sensitivity to these compounds is low (i.e., it takes a high concentration to detect them). Thus, although ketones are common byproducts of decomposition, they are not major odorants in themselves at composting sites.
5.3 Aldehydes
Several aldehydes inherently form as organic matter decomposes. The most recognizable aldehyde is formaldehyde, which is known by its penetrating medicinal odor. Acetaldehyde is ubiquitous compound characterized by a variety of odor descriptions including green, fruity, suffocating and garbage-like (Miller, 1993; Goldstein, 2002). Its character changes
6 References.
Brant, R.C., Elliott H.A., 2004. Odor Management in Agriculture and Food Processing in Pennsylvania, Pennsylvania State University, University Park, PA. https://www.agriculture.pa.gov/Plants_Land_Water/StateConservationCommission/OdorManagementProgram/Pages/default.aspx.
CalRecycle. 2007. Comprehensive Compost Odor Response Project (C-CORP). California Department of Resources Recycling and Recovery (CalRecycle). https://www2.calrecycle.ca.gov/Publications/Details/1241.
Chiumenti, A., R. Chiumenti, L.F. Diaz, G.C. Savage, L.L. Eggerth and N. Goldstein, 2005. Modern Composting Technologies, The JG Press, Inc. Emmaus, PA.
Das, K.C., 2000 Odor Related Issues in Commercial Composting, Workshop presented at the Y2K Composting in the Southeast Conference & Expo, October 2000, Charlottesville, VA.
Engineering Toolbox, (2006). Vapor Pressures common Liquids. [online] Available at: https://www.engineeringtoolbox.com/vapor-pressure-d_312.html [Accessed 26/09/2021].
Epstein, E., 1997. The Science of Composting. Technomic Publishing Company, Penn.
Goldstein, N., 2002. Getting to Know the Odor Compounds, BioCycle, Vol. 43(7).
Haug, R.T., 1993. The Practical Handbook of Compost Engineering. Lewis Publishers, CRC Press, Boca Raton, Florida.
Mendrey, K. 2014. The Compost Odor Wheel. BioCycle, February 21, 2014. https://www.biocycle.net/the-compost-odor-wheel/
Miller, F.C., 1993. Minimizing Odor Generation. In Science and Engineering of Composting. Ohio State University, pp. 219.
Walker, L.P. and J.M. Gossett, 1999. Controlling Odors and Waste Stabilization in Composting Systems Through Process Design, Analysis and Monitoring. The New York State Energy Research and Development Authority. Albany, NY.
Williams T. O., and F.C. Miller, 1993. Composting Facility Odor Control Using Biofilters: Minimizing Odor Generation. In Science and Engineering of Composting. Ohio State University.
Willmink, T.R., and R.G. Diener, Handbook for Commercial and Municipal composting in West Virginia. (2001).
***